Health
FDA Grants Full Approval To Leqembi, The First Alzheimer’s Drug Shown To Slow Cognitive Decline
(CTN NEWS) – The Food and Drug Administration (FDA) has endorsed the intravenous (IV) drug Leqembi for patients with mild dementia and early Alzheimer’s disease symptoms.
This marks the first time a medication targeting the underlying disease process of Alzheimer’s has demonstrated clinical benefits.
FDA Confirms Leqembi’s Effectiveness in Slowing Cognitive Decline in Alzheimer’s Patients
The FDA’s confirmation comes after reviewing data from a comprehensive study involving 1,800 patients, which showed that Leqembi modestly slowed memory and thinking decline by approximately five months compared to a placebo.
Teresa Buracchio, acting director of the Office of Neuroscience in the FDA’s Center for Drug Evaluation and Research, expressed her satisfaction with the results, stating that the confirmatory study verified the drug’s safety and effectiveness in treating Alzheimer’s disease.
Leqembi, developed by Japanese drugmaker Eisai, received conditional approval from the FDA in January based on early indications that it effectively cleared the sticky brain plaque associated with the disease.
FDA Approval Brings Hope, But Caution Remains Due to Possible Risks
However, the prescribing information for Leqembi carries a warning about potential side effects, including brain swelling and bleeding, which can occur in rare cases.
Similar side effects have been observed with other plaque-targeting Alzheimer’s drugs.
The FDA’s full approval of Leqembi has been eagerly anticipated by Alzheimer’s patients, advocates, and Medicare officials.
Medicare had initially decided not to cover the routine use of Leqembi until it received full FDA approval due to concerns about the financial implications of new plaque-targeting drugs for the program’s coverage of millions of seniors.
Leqembi is priced at approximately $26,500 for a year’s supply of IVs administered every two weeks.
Following the FDA’s decision, Medicare administrator Chiquita Brooks-LaSure confirmed that Medicare will now cover the drug, but additional requirements will be put in place.
Medicare recipients receiving Leqembi will need to be enrolled in a federal registry to monitor the drug’s real-world safety and effectiveness.
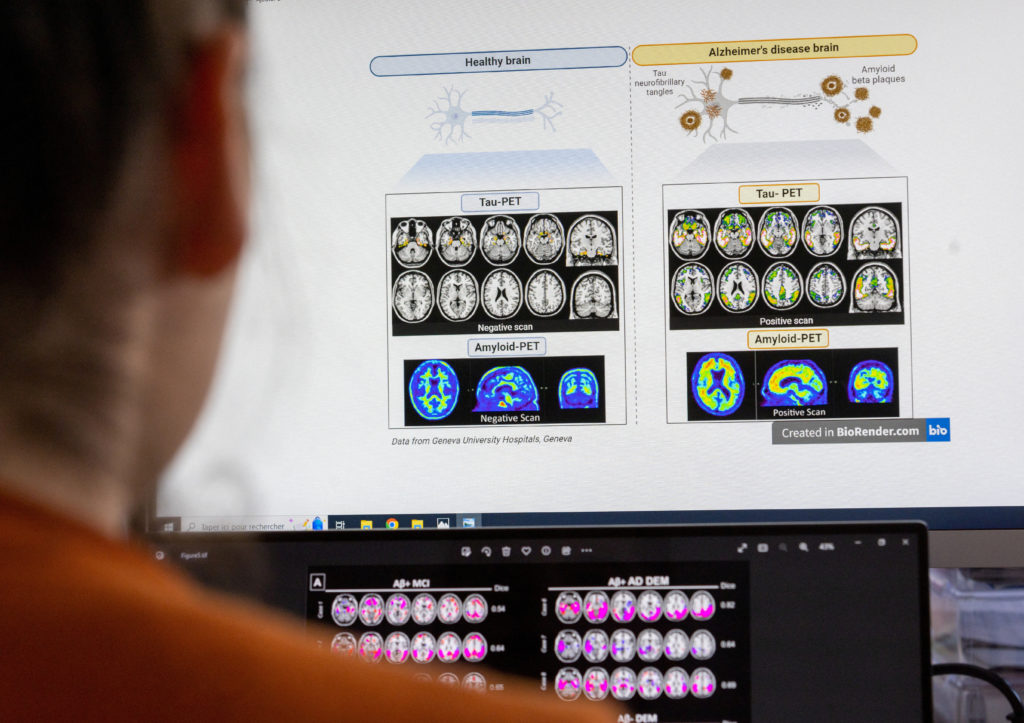
A scientist looks at scans of grains at the Memory Centre at the Department of Readaptation and Geriatrics of the University Hospital (HUG) in Geneva, Switzerland, June 6, 2023. REUTERS/Denis Balibouse
Implementation Challenges and Future Potential Surrounding Leqembi
Hospitals and medical clinics have cautioned that the process of initiating patients on Leqembi may take time. Doctors must confirm the presence of the targeted brain plaque before prescribing the drug.
Nurses require training to administer the drug, and patients must undergo repeated brain scans to monitor for potential side effects. These additional services carry extra costs for hospitals, beyond the cost of the drug itself.
Eisai estimates that around 100,000 Americans could be diagnosed and eligible for Leqembi by 2026. The drug is co-marketed with Biogen, based in Cambridge, Massachusetts.
While the delay in cognitive decline provided by Leqembi may be subtle and potentially unnoticed by patients and their families, federal health advisers deemed the difference to be meaningful.
RELATED CTN NEWS:
Walt Nauta Pleads Not Guilty To Charges Of Concealing Classified Documents
10 Men Sentenced To 10-Year Jail Terms For Lynching Muslim Man In India