News
Mother in Thailand Says Her Babies Eyes Turn Blue After COVID-19 Treatment
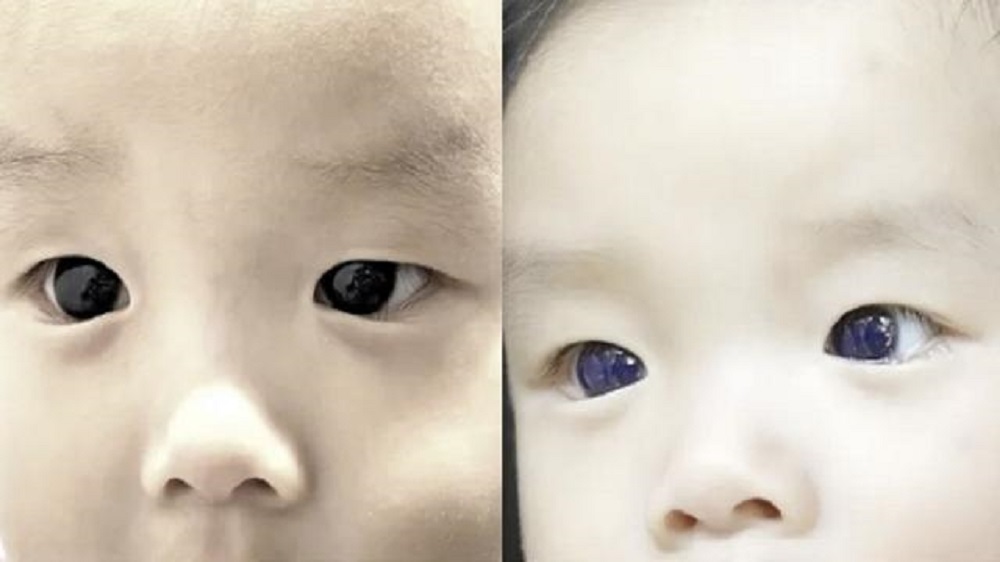
A 6-month-old boy in Thailand reportedly had his dark brown eyes turned bright blue after receiving a routine COVID-19 medication Favipiravir, in an uncommon case of a medical adverse effect.
The baby, who resides in Thailand, was identified with COVID-19 after suffering from a fever and a cough for a day, according to the medical journal Frontiers in Paediatrics. He had been on Favipiravir for three days, and it had helped improve his COVID symptoms.
However, only 18 hours after starting the drug, the child’s mother noticed a change in the colour of her baby’s eyes, which went from dark brown to vivid blue.
After noticing the colour shift, the doctor decided to discontinue the treatment, which returned to its natural brown shade five days later.
“There was no bluish discoloration in other areas such as skin, nails, or oral and nasal mucosa.” After three days of Favipiravir medication, the symptoms improved. Because of Favipiravir-induced corneal darkening, the paediatrician advised the patient to discontinue medication.
The cornea restored to normal colour on Day 5 after discontinuing the medication, according to the authors of the medical report.
It should be noted that Favipiravir, the antiviral drug approved by the Thai Ministry of Public Health in 2022, is meant for children with mild to moderate COVID-19 symptoms.
In a similar incidence in 2021, a 20-year-old male in India reported the onset of an uncommon adverse reaction to the medication. On the second day of Favipiravir medication, his previously dark brown eyes had turned a vivid blue colour.
The medical experts concluded the research by saying, “this case report highlights an unusual adverse effect of Favipiravir therapy in the youngest known patient receiving the drug for the treatment of SARS-CoV-2 infection.”
While Favipiravir is presently the basis of oral antiviral treatment for children with COVID-19, its safety profile in developing children is unknown.”
This case report emphasises an uncommon side effect of Favipiravir medication in the youngest known patient receiving the medicine for the treatment of SARS-CoV-2 infection, according to the report.
While Favipiravir is presently the basis of oral antiviral treatment for children with COVID-19, its safety profile in developing children is unknown.
As a result, monitoring the long-term safety of Favipiravir in paediatric patients is critical. Although the reported adverse event is rare, it should be taken carefully and closely monitored in future cases. Further research is required to assess the frequency of these adverse effects and their possible long-term effects on corneal health.





























