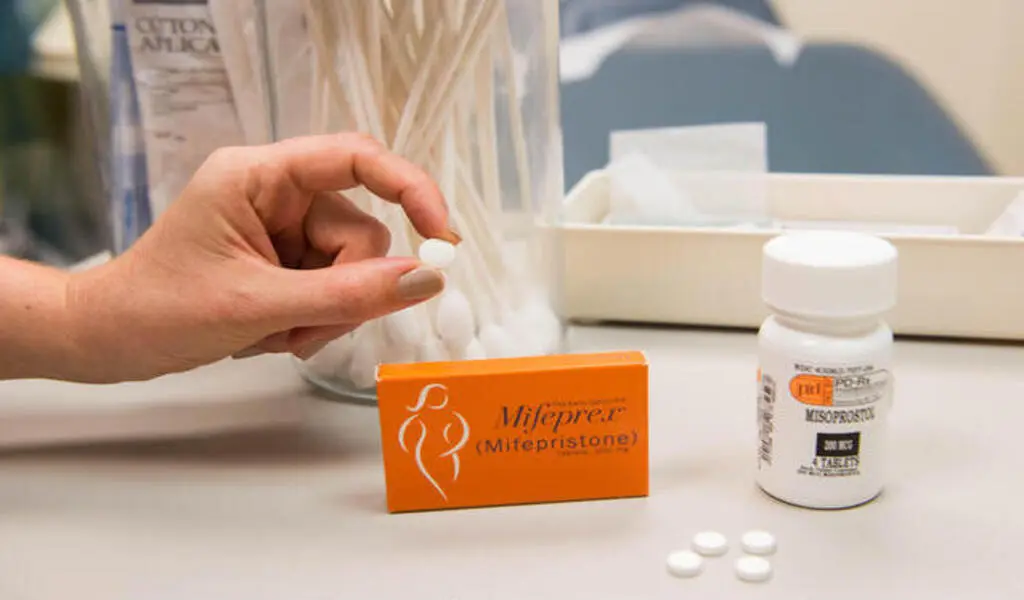
(CTN News) – The availability of the abortion pill mifepristone could be drastically affected by upcoming decisions from two federal judges.
This event took place in the state of Washington in the United States. Judge Thomas Rice of the District Court is considering whether or not to overturn federal regulations on mifepristone that make access difficult even in states where abortion is allowed.
He is also considering whether to issue an injunction preventing the FDA from taking steps to remove the drug from sale or limit access to it.
Judge Matthew Kacsmaryk of the United States District Court for the Southern District of Texas is debating whether to order the FDA to remove mifepristone from sale in the United States.
In November, pro-life medical groups filed a lawsuit against the FDA to reverse its drug clearance more than two decades ago.
Judges in Washington and Texas may issue rulings on mifepristone that conflict
The FDA and the Democratic attorneys general who launched the lawsuit challenging the FDA presented their cases to Judge Rice in Spokane on Tuesday. Less than an hour was spent on the whole meeting.
Earlier this month, Kacsmaryk heard arguments in the Texas case and promised to issue an order as quickly as possible. Kacsmaryk and Rice served in the Obama and Trump administrations and were chosen by their respective presidents.
We were well aware of the situation in Texas by the time we filed our complaint. “That’s just the legal world we’re living in,” said Washington State Attorney General Bob Ferguson, who is heading the case to keep mifepristone on the market and increase access to the drug.
Two federal district courts have the potential to issue conflicting decisions on the abortion pill, adding further confusion to the existing web of state-by-state laws on mifepristone.
Mifepristone cases could lead to Supreme Court review of abortion rights
In addition, the cases bring up the possibility that the Supreme Court will get involved in the growing number of lawsuits challenging the legality of the most prevalent abortion procedure in the United States.
In the event of a split decision regarding what the FDA should do, the case will almost certainly be sent to the United States.
Professor at Harvard Law School and former civil division attorney at the Justice Department Glenn Cohen emailed his thoughts on the Supreme Court. Cohen submitted a brief advocating for the FDA’s approval of mifepristone in the Texas case.
According to Ferguson, the plaintiffs in the Washington case requested the judge to increase and safeguard mifepristone access in the 18 jurisdictions involved (17 states and DC).
Medical groups opposed to abortion have asked a court in Texas to ban the sale of abortion pills across the country.
Ferguson said that the judge in Washington could still issue an order that at least protects access in the 17 states and D.C. if the judge in Texas rules first and orders the FDA to remove mifepristone from the market.
According to Ferguson’s statement, the case will be preserved in Washington state, and the plaintiff states that the federal court in Washington will be making a finding on Washington. There would be conflicting judicial decisions, but this can be resolved on appeal.
You might find that in some states, it is unavailable, while in others, it is readily accessible. It’s conceivable for any of those to occur. But, as Ferguson pointed out, “a lot of rides on the way these judges pen these rulings.”
Since its approval in 2000, mifepristone has been subject to government monitoring program restrictions, but these have been loosened over time by the Food and Drug Administration.
In January, it permitted mifepristone to be shipped to patients rather than requiring them to pick it up in person. In addition, the FDA finally authorized the pill’s sale in retail shops.
However, there are still some limitations imposed by the organization. Patients must acquire a prescription from a doctor certified under the federal monitoring program and sign a form outlining the risks of mifepristone. To give the medication to the patient, pharmacies must also be accredited through the program.
Attorney General Ferguson and his colleagues have asked the Washington state court to lift the ban.
Mifepristone use at the center of legal battles in several states
The 17 states are as follows: Arizona, Colorado, Connecticut, Delaware, Illinois, Michigan, Nevada, New Mexico, Oregon, Rhode Island, Vermont, Hawaii, Maine, Maryland, Minnesota, Pennsylvania, and Washington.
In their lawsuit, the attorney’s general told the court that the bill “only serves to make it harder for doctors to prescribe, harder for pharmacies to fill, harder for patients to access, and more burdensome for the Plaintiff States and their health care providers to dispense.”
Whether or not the Biden administration would challenge a ruling requiring the FDA to lift mifepristone restrictions was an issue Cohen said the Washington lawsuit raises.
Related CTN News:
Google Search For “Lance Reddick Cause of Death” Hits 2 Million