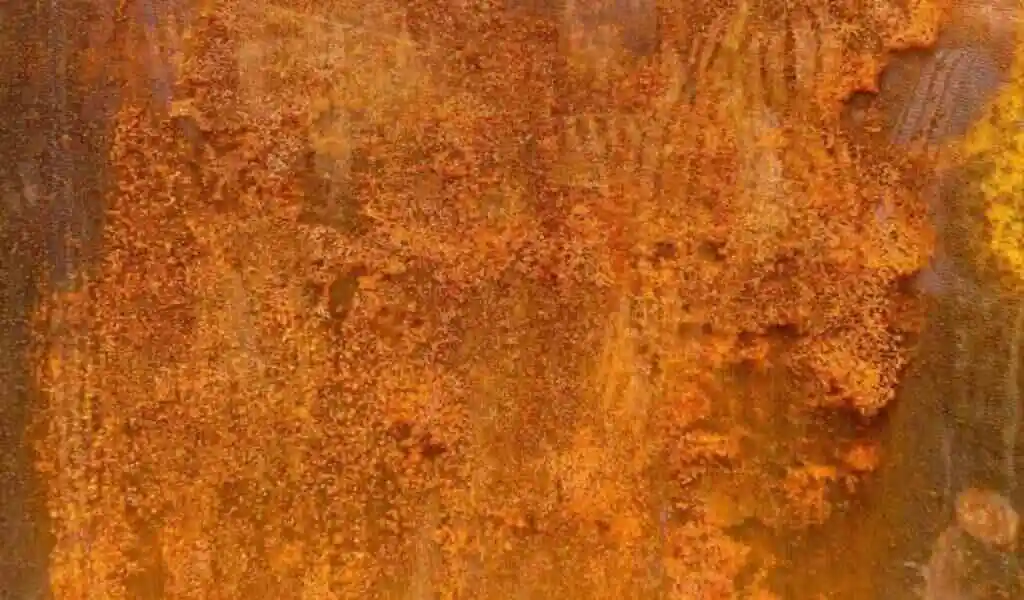(CTN News) – A common corrosion misconception is that iron rusts only when it comes into contact with oxygen and water. It is also possible for certain bacteria to decompose iron anaerobically, a process known as electrobiocorrosion.
Angewandte Chemie reports that the sediment-dwelling bacterium Geobacter sulfurreducens uses electrically conductive protein threads for this purpose.
In a positive feedback loop, they produce magnetite from iron, which further encourages corrosion.
Bacterial biofilms cause microbial metal corrosion, which is more costly than all other biofilm-related damage combined. It is common for electrobiocorrosion to occur as a result of bacteria found in river sediments, such as the anaerobic genus Geobacter.
Instead of drawing energy from atmospheric oxygen for respiration, Geobacter utilizes electrons transferred from iron to form magnetite. Until recently, the mechanism by which Geobacter corrodes iron metal remained a mystery.
As a result of their investigation, Dake Xu and colleagues from Northeastern University in Shenyang, China, have been able to determine the exact mechanism of electrobiocorrosion.
It was thought that electrically conductive pili, thin filaments that grow from bacteria, might play a significant role in this process. Geobacter forms “e-pili” from conductive proteins, which function like electric wires and conduct electricity. It was unclear whether the e-pili could directly withdraw electrons from metal surfaces before this study.
Two strains of Geobacter were grown on a stainless-steel surface until biofilms developed in order to prove the team’s suspicions, namely direct electron withdrawal.
In one strain, e-pili were formed from conductive proteins, while in the other strain, more conductive proteins were used to form pili.
Compared with the bacterial strain that grew e-pili on the steel plate, the bacterial strain that grew e-pili performed significantly better. The fungus grew faster and made deeper pits in the metal, demonstrating how much metal it was consuming. As a result of the oxidation of iron, the team measured a current.
Scientists concluded that bacteria with e-pili formed an electrical connection with metal. Using e-pili, bacteria located farther away in the biofilm, and not in direct contact with the metal, were also able to acquire electrons.
As magnetite is formed during the of iron and conducts electricity, the team studied its impact on microbial corrosion. Magnetite contributed to an increase in Geobacter growth in the biofilm, but it also contributed to an increased current measured at the metal’s surface.
As the team points out, the discovery that magnetite, a common product, facilitates electrobiocorrosion has significant implications for corrosion.
They recommend taking into account the tendency of materials to form magnetite in future attempts to improve corrosion protection.
SEE ALSO:
PIA’s Inaugural Dubai To Skardu Flight Marks A Milestone In Accessibility And Travel Efficiency
Newlywed Bride Seeks Refund Over Photographer’s Alleged Involvement With New Husband