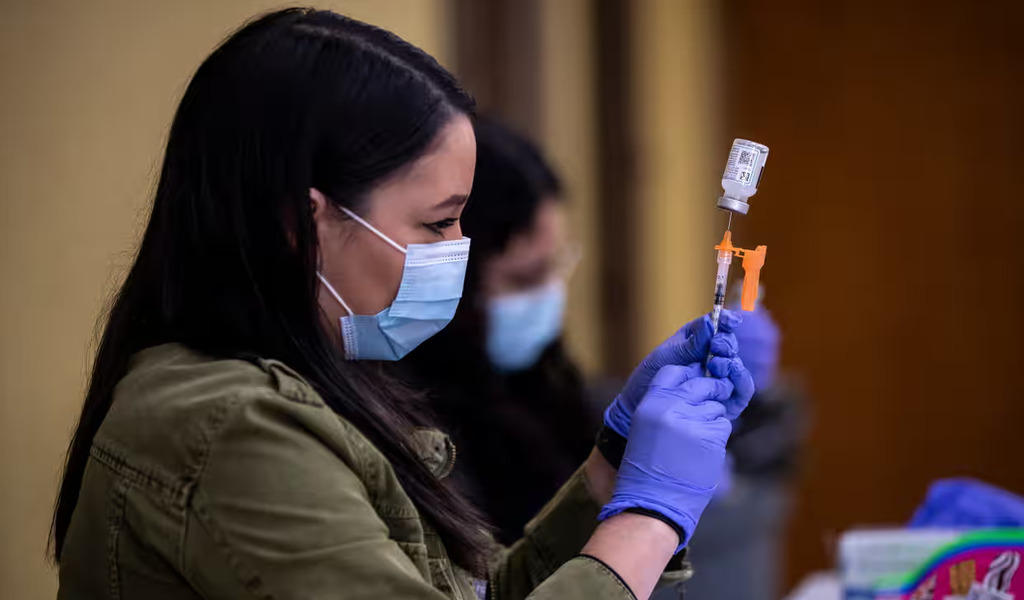
(CTN NEWS) – A revamped single-dose COVID “booster” vaccine is anticipated to become available at pharmacies, coinciding with the onset of the cold and flu season.
In the latest development, the Food and Drug Administration (FDA) has granted emergency use authorization for these new booster shots.
Furthermore, a committee of experts at the Centers for Disease Control and Prevention (CDC) has endorsed the recommendation that almost all individuals in the United States should receive this booster shot.
When should Americans consider obtaining the new COVID vaccine, and how crucial is it given the prior vaccinations and infections? Below, we delve into the latest recommendations from health agencies.
What Did the FDA Approve?
The FDA granted authorization for updated versions of the Pfizer and Moderna mRNA vaccines. As COVID, much like the flu, keeps mutating, pharmaceutical companies have devised a booster shot targeting one of the prevailing strains this season, the XBB 1.5 variant.
This autumn marks the first opportunity for adults to receive vaccines for influenza, respiratory syncytial virus (RSV), and COVID-19 simultaneously, all of which safeguard against commonly circulating respiratory illnesses.
This year’s COVID booster specifically addresses the XBB 1.5 variant.
Who Should Receive the Booster?
When the FDA authorizes a new vaccine, the CDC issues recommendations regarding who should receive it and when. In essence, the agency has advised this booster for everyone over six months old, with a few exceptions.
Individuals recently infected with COVID-19 can wait for three months before getting the updated booster, and those who were recently boosted should wait two months before receiving the new shot.
The expert panel at the agency places particular emphasis on specific groups getting the booster.
Dr. Peter Hotez, a vaccinologist and dean of the National School of Tropical Medicine at Baylor College of Medicine in Houston, Texas, states, “The most important are those at greatest risk for severe illness.”
This category includes those over 65, individuals with conditions like obesity, diabetes, high blood pressure, chronic lung or heart disease, and those who may be immunocompromised, such as individuals taking immune-suppressing medication and pregnant women.
Why Should I Get Another Shot If I’m Already Vaccinated or Recovered from COVID?
Even though a significant portion of the US population has acquired immunity through vaccination or prior infection, the likelihood of contracting COVID again increases over time as new variants emerge and vaccine-induced immunity wanes.
The agencies aim to prevent severe illness, the type that might necessitate hospitalization or result in death, by providing updated vaccines.
Additionally, data suggests that receiving a COVID-19 vaccine can reduce the risk of developing “long COVID,” where symptoms like fatigue and brain fog persist for months. Consequently, the agencies have recommended a broad vaccination campaign for the American public.
“I’ll be first in line,” says Dr. Hotez, expressing support for the CDC’s comprehensive recommendation, “Even though the number of hospitalizations will not reach anywhere near the levels seen in 2021 and 2022.
In terms of making an individual health choice, I think there’s still vulnerability.”
How Long Does the Immunity from the New Shot Last?
Peak protection should be achieved a couple of weeks after the booster, and it should last for a few months. After this period, immunity diminishes, either due to new variants in circulation or declining vaccine efficacy.
The exact duration is challenging to ascertain based on the available data.
Is There Universal Agreement Among Experts on the CDC’s Recommendation?
While a CDC expert panel endorsed the new recommendations, not all experts concur with the strategy.
Dr. Paul Offit, a virology and immunology expert at the Children’s Hospital of Philadelphia and a member of the FDA’s vaccine expert panel, argues that the government should prioritize the most vulnerable groups.
In 2022, only 43% of individuals over 65, a high-risk group for hospitalization due to COVID, received a booster. Dr. Offit contends that this specific group should be the focal point of a booster campaign.
He says, “My feeling on this, and it’s a sentiment shared by health advisors in many other countries, is the goal of this vaccine is to prevent serious illness. Then, the question becomes who is most likely to suffer serious illness? Who is at greatest risk?”
According to CDC data, the highest-risk groups include those over 75 with underlying health conditions (such as those mentioned earlier) and the immunocompromised.
With minor exceptions, like nursing home workers and those residing with the immunocompromised, Dr. Offit asserts that the “best strategy is to target those of greatest risk.”
Will I Have to Pay for the New Vaccine?
Most likely not. Health insurance should cover the cost of the booster dose for the majority of Americans. For the approximately 27 million Americans without health insurance, the federal Bridge Access Program will cover vaccine expenses through 2024.
For those who choose to pay out of pocket, Pfizer and Moderna have established the list prices for their vaccines at $120 and $129, respectively.
RELATED CTN NEWS:
Golden Jubilee Celebration: King Carl XVI Gustaf’s 50 Years On The Swedish Throne | Things To Know
Japan’s PM Kishida Appoints Record Number Of Women In Cabinet Reshuffle Amid Ratings Slump
Pakistani Fashion Model Erica Robin Makes History As The First ‘Miss Universe Pakistan’