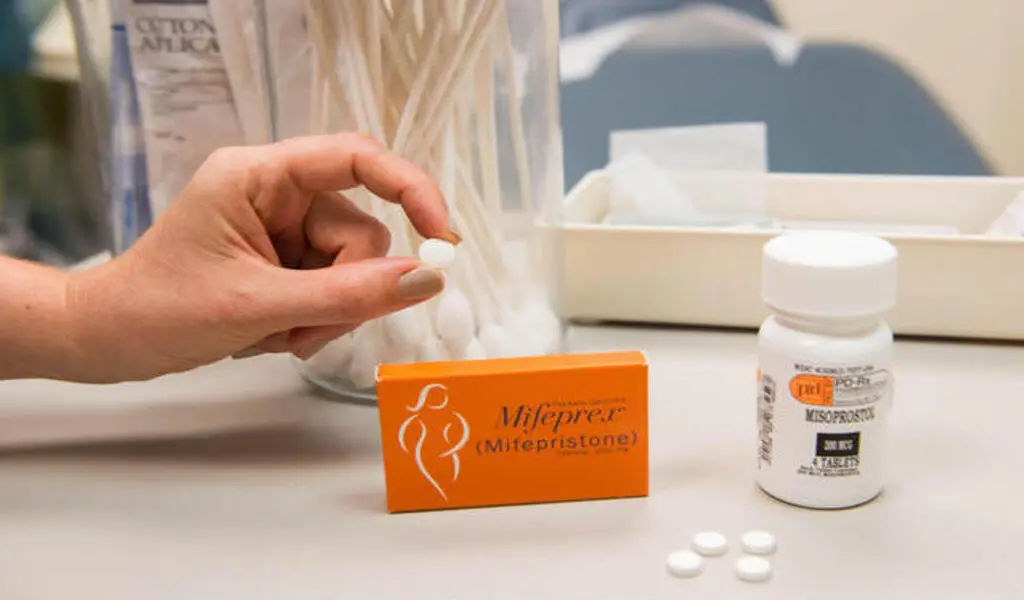
(CTN News) – The abortion pills mifepristone may be obtained by persons with a prescription from accredited pharmacies according to a decision by the US Food and Drug Administration.
To stop a pregnancy, mifepristone may be used with the drug misoprostol. Before this change, a licensed healthcare professional was the only one who could order, prescribe, and distribute these medications.
The FDA said it would no longer enforce a requirement requiring consumers to get the first of the two medications in person at a clinic or hospital during the Covid-19 epidemic and permitted the tablets to be supplied over mail.
The medicine is marketed under the brand name Mifeprex by Danco Laboratories, and as of Tuesday, the in-person requirement has been permanently eliminated.
Those with a prescription from a licensed physician may get the medications straight from pharmacies that have obtained the necessary certification.
On Tuesday, the FDA’s website revealed the new details.
This modification is crucial to increasing access to medication abortion services and will give healthcare providers another way to offer their patients a safe and effective option for terminating early pregnancy, according to a statement from Danco.
People across the country are currently having difficulty obtaining abortion care services.
State-specific regulations differ; however, the drugs may be used for up to 11 weeks from the first day of the last menstruation.
Some states allow telehealth prescriptions, or a person might fly to a state where abortion is permitted to get the pills.
The Guttmacher Institute, a research organization that promotes abortion rights, claims that medication abortion is used in more than half of abortions in the US, surpassing surgical procedures for the first time in 2020.
CVS, a national pharmacy chain, said in a statement on Tuesday that it is studying the revised specifications.
To ascertain the requirements to dispense in states that do not restrict the dispensing of medications prescribed for elective termination of pregnancy, we are reviewing the FDA’s updated Risk Evaluation and Mitigation Strategy (REMS) drug safety program certification requirements for mifepristone.
Walgreens said that it is examining the modifications.
The FDA’s action comes after the Justice Department issued a new legal opinion stating that federal law permits the US Postal Service to deliver abortion drugs.
The Biden Administration believes that this could help protect access to abortion in states that have enacted bans as a result of the Supreme Court’s decision to overturn Roe v. Wade.
The 1873 Comstock Act “does not prohibit the shipping of certain medications that may be used to cause abortions if the sender lacks the intent that the receiver of the drugs would use them illegally,” according to the Office of Legal Counsel opinion December 23, 2022.
The opinion continued, “The mere mailing of such drugs to a particular jurisdiction is an insufficient basis for concluding that the sender intends for them to be used unlawfully because there are numerous ways in which recipients in every state may lawfully use such drugs, including to produce an abortion.
The USPS asked the OLC for guidance after Roe v. Wade was overturned last year over whether federal law prohibited the shipment of mifepristone and misoprostol.
Attorney General Merrick Garland promised in a statement after the Supreme Court decision to collaborate with the FDA and other federal agencies to defend access to these medications, which some states have attempted to outlaw.
According to Garland, “States may not restrict Mifepristone based on disagreement with the FDA’s expert judgment regarding its safety and effectiveness.”
Related CTN News:
Plan B Label Clarifies ‘Morning-After’ Pill Doesn’t Cause Abortion