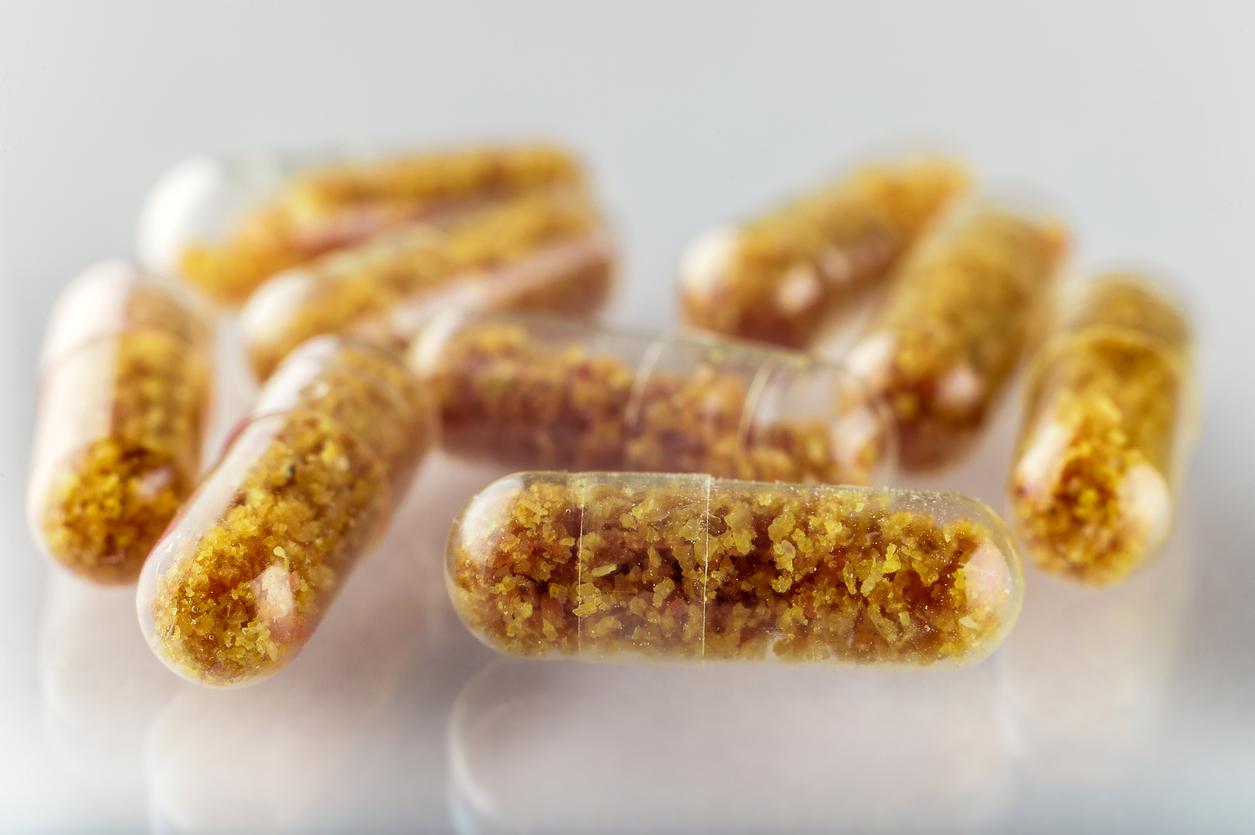
(CTN News) – On Wednesday, U.S. health officials approved the first tablet manufactured from beneficial bacteria found in human feces to treat deadly gut infections, making it simpler to carry out so-called faecal transplants.
To help patients, certain medical practitioners have used stool-based procedures for more than ten years. The new treatment from Seres Therapeutics offers a more straightforward, thoroughly tested alternative.
The capsules were approved by the Food and Drug Administration for use by adults age 18 and older who run the risk of contracting Clostridium difficile, a bacterium that can result in severe nausea, cramps, and diarrhoea.
When C. diff recurs, it is more hazardous and causes 15,000–30,000 fatalities annually
Antibiotics can eliminate it, but they also kill the beneficial bacteria that exist in the stomach, making the body more vulnerable to further infections.
For patients who have already undergone antibiotic therapy, the new pills have been approved.
In order to restore the gut’s normal balance and avoid reinfections, several doctors started reporting success with faecal transplants more than ten years ago. These procedures use stool from a healthy donor.
Last year, a competing pharmaceutical company, Ferring Pharmaceuticals, submitted the first medication in pharmaceutical-grade form to the FDA for approval.
However, like most of the original methods, the company’s product needs to be given through the rectum.
Cambridge Massachusetts-based Seres will market its drug as a less invasive option
The medication, which will be offered under the trade name Vowst, is administered as a course of four daily pills over the course of three days.
The pharmaceutical industry has spent years studying the microbiome, or the population of bacteria, viruses, and fungi that dwell in the gut, and two recent FDA approvals are the results of that research.
Currently, a network of stool banks that have appeared in medical facilities and hospitals around the nation offer the majority of faecal transplants.
While the demand for donations from stools banks is anticipated to decline as new FDA-approved alternatives become available, some intend to continue operating.
The largest stool bank in the United States, OpenBiome, has stated that it will continue to assist patients who are not qualified for FDA-approved medicines, such as youngsters and adults with illnesses that are resistant to therapy.
Since 2013, it has provided more than 65,000 stool samples for C. diff patients.
Dr. Majdi Osman, the organization’s chief medical officer, stated that “OpenBiome is committed to maintaining safe access to ‘faecal transplantation’ for these patients as a vital last line of defence.”
The usual stool treatment from OpenBiome costs less than $1,700 and is typically shipped as a frozen solution within days of placing an order.
In a statement released on Wednesday night, Seres omitted to include the cost of its capsules.
Eric Shaff, the company’s chief executive officer, stated in an interview conducted prior to the release that “we want to make the commercial experience for physicians and patients as easy as possible.”
“In our opinion, one of the aspects of the value we’re delivering is ease of administration.”
Thousands of capsules can be made from each stool sample
Together with Swiss food juggernaut Nestle, Seres will co-market the treatment, sharing earnings in the process. With regard to the FDA approval, Seres will get a $125 million milestone payment from Nestle.
The FDA has faced regulatory challenges overseeing the nascent U.S. stool bank industry since it doesn’t typically regulate handmade items and practices used in medical offices.
The FDA initially cautioned consumers about the dangers of possible illnesses from faecal transplants as some people looked for dubious “do it yourself” techniques on YouTube and other websites.
Executives from Seres claim that their manufacturing process uses the same methods and tools for blood product and other biologic therapy purification.
The business begins with faeces that is given by a select group of donors who are examined for a variety of health issues. Additionally, dozens of potential viruses, illnesses, and parasites are examined in their stool.
The business then refines the samples to eliminate waste, isolate good bacteria, and eradicate any leftover microorganisms.
Common Side Effects
According to the business, it is a more effective procedure than current faecal transplants because thousands of capsules may be produced from each stool sample.
The FDA stated that the medicine “may carry a risk of transmitting infectious agents” in its statement of approval
Additionally, Vowst may contain food allergies, the government stated.
Based on a 180-patient research, the FDA approved the therapy because, compared to 60% of patients who got placebo tablets, over 88% of those receiving the capsules did not have reinfection after 8 weeks.
Constipation, diarrhea, and abdominal swelling were frequent adverse effects.