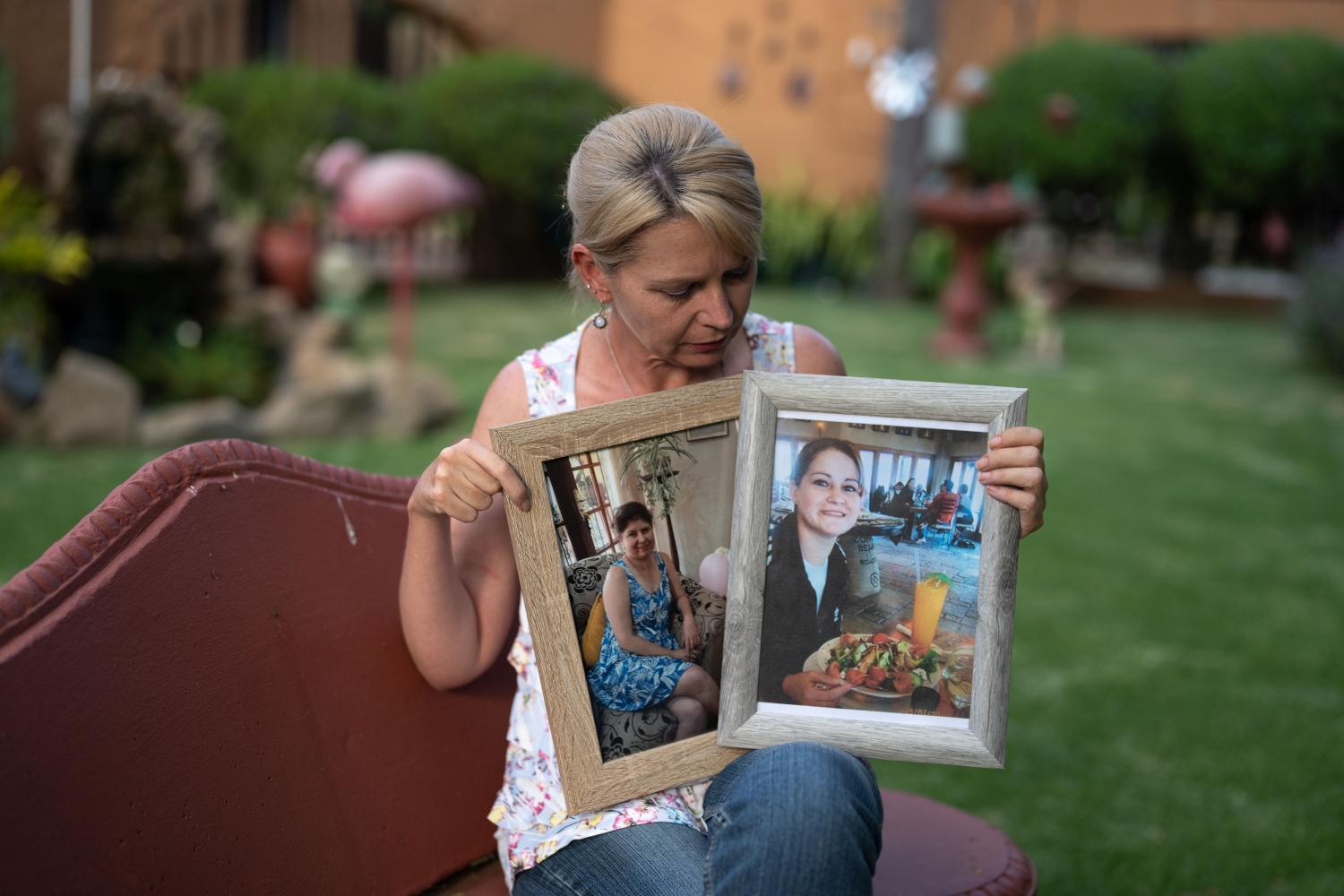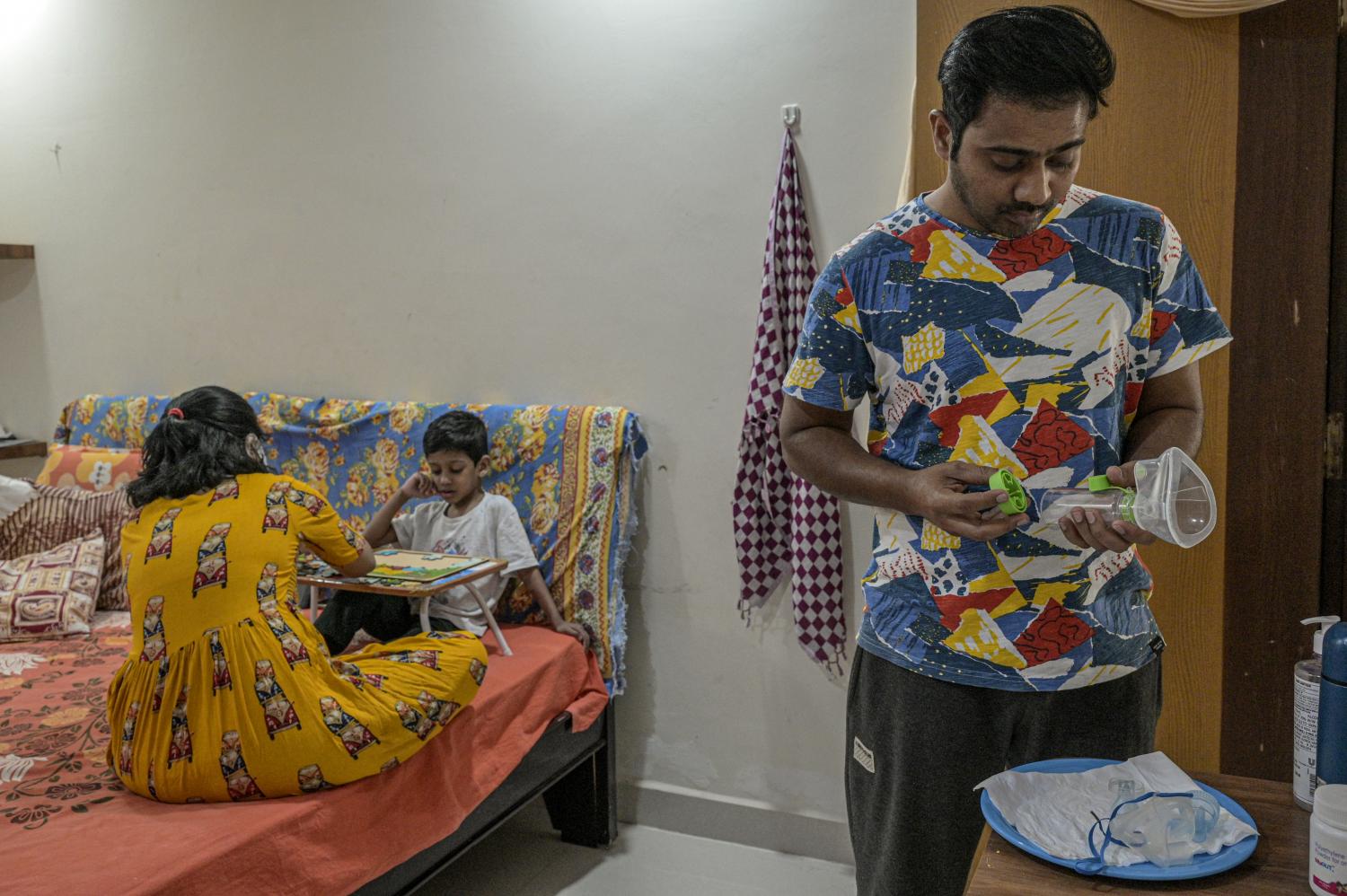When Seshagiri Buddana learned of a powerful new cystic fibrosis drug that was transforming lives in the United States and Europe, he was filled with hope that it could help his son, Hemanth, who had spent much of his childhood in a hospital bed. But the family couldn’t get the drug because they live in India.
Big Pharma drug maker, Vertex Pharmaceuticals, a large biotech company based in Boston, is not making it available in India or virtually anywhere in the developing world. The company is not trying to sell it, or allowing a local company to make it. Vertex is blocking potential generic competitors by seeking patents in numerous countries.
Hemanth died in December, a day before his ninth birthday and 18 months after he would have been eligible to get the drug, called Trikafta, had he lived in the United States.
Throughout much of Asia, Africa and Latin America, families like Hemanth’s are watching Trikafta transform the lives of tens of thousands of cystic fibrosis patients in wealthy nations but say they are blocked by the company at every turn in their efforts to get the drug themselves.
Trikafta, taken as three tablets a day, is the most powerful and widely used of Vertex’s four cystic fibrosis medications. With a list price of more than US$322,000 (10.9 million baht) annually in the United States, it is expected to cost millions of dollars over the course of a patient’s lifetime.
An analysis led by researchers in Britain found that a year’s supply of the drug could be manufactured at an estimated cost of just $5,700.
Vertex has reported more than $17 billion in sales for Trikafta since it was first approved in 2019.
Last week, a group of patients and their families in four countries on four continents initiated legal and regulatory steps to try to force their governments to override intellectual property protections and allow a low-cost generic version of Trikafta to be imported or made locally.
Under the process, known as compulsory licensing, generic makers would pay Vertex a royalty.
Three of the actions are in India, Ukraine and South Africa — where Vertex has been obstructing efforts to make the drug available, patients and families say. The fourth is in Brazil, where Vertex is trying to win coverage for the drug; the patients and families’ concern there is that the brand-name drug will be too expensive.
Cystic fibrosis is a genetic disease that damages the lungs and digestive system. Patients often die in early adulthood, but Trikafta is dramatically extending life expectancy.
“Every patient in the world has access to the internet and wants this drug,” said Christine Noke, a patient advocate in Turkey.
In theory, reaching patients in the developing world would bring in more revenue for a drug company. But some manufacturers resist making their drugs available in poorer countries at lower prices because doing so can erode their ability to charge more in high-income countries.
Rodrigo Rockenbach, 11, who lives with cystic fibrosis, uses a nebuliser as his parents look on in Curitiba, Brazil. Dado Galdieri/NYT
Vertex, which has a monopoly on transformative cystic fibrosis drugs, said it was pushing to increase access globally.
“Our teams are working every day to expand access to even more patients around the world through a range of routes, including in low-middle-income countries and low-income countries where access barriers are high due to challenging economic conditions and limited health care infrastructure,” said Heather Nichols, a spokeswoman for Vertex.
Ms Nichols said that Vertex has begun a “product donation programme” in low-income countries. She said the company has provided some form of access to at least one of its cystic fibrosis drugs in Brazil, Poland, Bulgaria, Estonia, Greece, Latvia, Slovakia, Slovenia, Romania and Oman. The company declined to specify which lower-resourced countries have access to Trikafta.
The genetic defect that causes cystic fibrosis is most common in people of Northern European ancestry, as are the specific mutations needed for Trikafta to work. The number of cystic fibrosis patients in developing countries who are diagnosed and eligible for the drug is unknown but believed to number in the thousands.
In India, a recent survey counted just 600 diagnosed cystic fibrosis patients. Counting India’s tens of thousands of patients who have not been diagnosed, some researchers estimate that India’s total cystic fibrosis population is higher than that in Europe.
While a minority of Indians with cystic fibrosis are believed to have mutations that make them eligible for Trikafta, the size of India’s population translates into huge numbers of patients who could benefit from Trikafta.
Hemanth Buddana, the Indian boy who died, was given therapies and antibiotics for his frequent lung infections, but there was little available in India to help him breathe or gain weight. Stuck in bed at home in Hyderabad, he taught himself to draw and to speak new languages.
A genetic test confirmed that he would be eligible for Trikafta, which has a US list price 20 times as much as the annual salary Seshagiri Buddana earns as an operations manager at Google. He joined other parents in pushing the Indian government to find a way to get Trikafta for their children. But there was no progress.
“They say it’s a miracle drug, but it’s not a miracle if it is not available to everyone who need it,” said Shwetha Sree, who also lives in Hyderabad. Her 5-year-old son, Vihaan, has cystic fibrosis — and the mutation that would make him eligible for the drug when he turns six, if he were to live in the US.
Since the fight over access to HIV treatment in sub-Saharan Africa in the early 2000s, some drug companies have agreed to sell their medicines at a profitable but significantly lower price in developing countries. The companies also sometimes work with a drug importer to sell the products in those regions.
There is also compassionate use, through which drug companies supply products to desperate patients in places where they are unauthorised. Vertex said that it has provided its medications free of charge to 6,500 patients worldwide that way.
The company declined to say specifically where it has provided the drugs that way and where it is still doing so.
A company can also agree to voluntary licences, allowing generic manufacturers to make and sell a drug in certain countries, typically in exchange for a royalty.
The Medicines Patent Pool, a United Nations-backed nonprofit that brokers that process by issuing sub-licences to generic manufacturers, said it has had no contact with Vertex.
“New drugs typically take longer to reach poorer countries. But frustration with Vertex’s failure to provide them with any form of access brought together cystic fibrosis drug patients online and led to a coordinated campaign for compulsory licensing”.
Governments are often reluctant to do compulsory licensing, which capital markets tend to view as an alarming crack in the wall of intellectual property protection. Still, even if governments refuse to issue a compulsory licence, the patient actions may pressure Vertex to make Trikafta available in those countries.
Cheri Nel, a 38-year-old investment banker in South Africa who has cystic fibrosis and is eligible for Trikafta, said that she had approached Vertex and suggested several ways the company could increase access and still safeguard its profit and intellectual property. She said she got nowhere and is now leading the action in South Africa.
“There’s a balance: You want to keep companies incentivised to investigate and do research and development,” she said. “But it does them no financial harm to let us import a generic because they’re not even trying to sell it.”
Vertex has not registered Trikafta with South Africa’s drug regulator, but the company said that it recently signed an agreement with a distributor there.
In many countries, Vertex has also been seeking patents that deter generic manufacturers from selling the drug there, according to patent filings viewed by The New York Times. The company has a running legal battle with Gador, one of several manufacturers in Argentina making lower-cost generic versions of Vertex’s drugs.
Argentina does not recognise Vertex’s intellectual property rights because the company has not joined the global treaty on patent protection. Gador’s scientists reverse-engineered the Vertex drugs and began to sell them to Argentine patients.
Then patients from foreign countries began to fly to Argentina to buy the drugs, which can cost as little as $18,000 per year using pesos exchanged on the black market.
Raphaelle Pereira, who lives with cystic fibrosis, is looking forward to the removal of the port-a-cath that was inserted for her frequent infections, in Curitiba, Brazil. Dado Galdieri/The New York Times
Until Vertex’s drugs, patients had few options, mainly palliative treatment to help them breathe a bit better, and if they could get one, a lung transplant. Vertex’s drugs addressed the underlying cause of the illness, preventing patients’ lungs from clogging in sludgy mucus.
Trikafta is stunningly effective at helping patients breathe better, keeping them out of the hospital and extending their lives. Patients and doctors say that the drug’s power becomes evident almost immediately.
Raphaelle Pereira, 22, had been waiting for years for a lung transplant in the Brazilian city of Curitiba. By 2021, her weight had dropped to 36 kilogrammes, and she no longer had the strength to walk to the bathroom. Family members sold property and scraped together $54,000 to purchase a two-month supply of Trikafta in the US.
“I took it for a couple of days, and then I just got up and said, ‘I think I’ll have a shower.’ My whole family was in shock,” she said. “A few days before I couldn’t even lift my arm.”
With the data on how the medication had changed her condition, Ms Pereira used a legal process to get Brazil’s public health system to buy a steady supply for her. She’s now working toward a career as a soccer commentator.