Regional News
Health Minister Defends Thailand’s Highly Disputed Vaccine Program
Thailand’s Public Health Minister has insisted authorities never stopped the private sector from importing Covid-19 vaccines as criticism escalates, even after the government confirmed that 10 million doses could be ordered by private hospitals.
Public Health Minister Anutin Charnvirakul said on Saturday that Prayut Chan-o-cha’s government fully supported plans by the private hospitals to buy “alternative vaccines”.
Presently Prime Minister Prayut Chan-o-cha’s government only procured China’s Sinovac Biotech and Oxford’s AstraZeneca vaccines. These are the only two vaccines Gen Prayut’s government procured for its free jab program over the next two years. Criticism has been raised of why only these two when other effective vaccine are more readily available.
Critics of Gen Prayut’s governments vaccination program question why authorities never sought alternate vaccines and allowed the private sector to procure the vaccines to better protect Thai citizens.
Private hospitals are now anticipating substantial demand from a large segment of the population who are willing to pay rather than having to wait up to 2 years for free vaccine shots. The private sector have been looking for ways to order alternative vaccines, especially those made by Pfizer, Moderna, and Johnson & Johnson.
Alternatives to governments vaccine program
On Saturday to ward off critics Mr Anutin said his ministry had in fact been looking for more choices. “If the private sector can deal with Pfizer, it can register with the FDA [Food and Drug Administration],” he said.
He said that his ministry had been in talks with other vaccine manufacturers but they all set minimum orders and specific delivery times that didn’t suit our requirements, without revealing the specifics.
Thailand has approved vaccines from three manufacturers. In addition to the two already ordered, J&J was also registered, he said.
“We contacted J&J but were told the vaccine could be delivered by the end of this year at the earliest, which coincides with [the delivery of] AstraZeneca. We won’t be needing it that much by then,” said Mr Anutin.
The minister explained that the government’s procurement plan also took into account the mutation factor.
“We won’t buy in large numbers — we will only buy as many as we can administer — to keep the inventory at a minimum.” He added it was imperative that the country had a “plan B” in case the virus mutated further.
“We don’t want to be sitting on a huge pile of useless vaccines that can’t keep up with the virus,” he said.
Slow pace of vaccinations
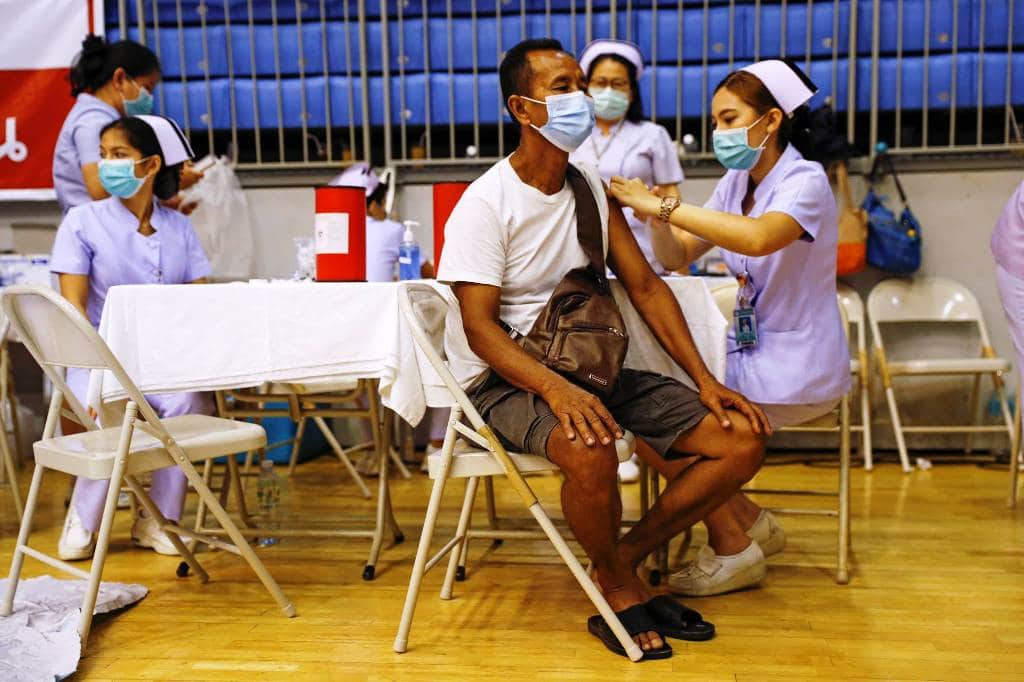
Mr Anutin’s comments followed criticism about the slow pace of vaccinations, especially in light of the arrival of a third wave last week. Hundreds of people have been infected from entertainment venues in Bangkok.
As of Saturday, Thailand ranked 124th out of 154 countries in the percentage of adults (0.4%) receiving a first vaccine dose, according to data compiled by The Economist.
Mr Anutin was not the first to try to quell public frustration about the slow pace of vaccine process. Furthermore the perception that the Gen Prayut’s government had been reluctant to allow Thais to obtain vaccines from other manufacturers.
AstraZeneca has contracted Siam Bioscience, a local company owned by His Majesty the King, to produce its vaccine for Thailand and the rest of Asean. The local facility is scheduled to deliver the first lot for local use by June.
Sinovac Biotech, meanwhile, is 15% owned by the Thailand-based conglomerate Charoen Pokphand group owned by the Chearavanont family. One of five billionaire families in Thailand that backed Gen Prayut as Prime Minister and made a fortune during the pandemic.
On Saturday, Prime Minister Prayut Chan-o-cha said local production of the vaccine by Siam Bioscience was progressing satisfactorily. “Everything has gone according to plan. It’s now in the process of quality checking at laboratories in the United Kingdom and the United States,” he said.
Furthermore an additional one million doses of China’s Sinovac vaccine arrived at Suvarnabhumi Airport on Saturday. Health officials would be distributed nationwide as soon as possible.
The shipment was the third lot, after 200,000 doses in late February and 800,000 the following month, bringing the total of China’s Sinovac vaccine to 2 million.
No-fault compensation
Last week, Dr Nakorn Premsri, director of the National Vaccine Institute, clarified “misunderstandings” about vaccine imports. He said all vaccines needed to be registered before they could be used. Some companies, including Pfizer, Moderna, AstraZeneca and Johnson & Johnson, have offices in Thailand that can act on their behalf.
“If their representatives here do not apply to register them, no one can do it for them,” he said. Other manufacturers with no local subsidiaries must appoint authorised representatives and submit documents for registration.
He cited Sinovac as an example, saying the Chinese company had the Government Pharmaceutical Organization represent it. Moderna has asked Zuellig Pharma in Thailand to act on its behalf.
“Companies seeking vaccines from other manufacturers must contact them directly and the government has never blocked such a move,” he said.
Some companies, he said, sold directly to the government, including Pfizer, AstraZeneca and Johnson & Johnson. As the vaccines are new for emergency use only. They all want the government to accept the “no-fault compensation” scheme, which prevents suits for civil damages in cases of severe side effects.
Liberalization of vaccine rules
Discussing supplies, Dr Nakorn said most well-known vaccines could be delivered in the fourth quarter of this year at the earliest and only in limited numbers. The exceptions are Sinovac, which has been gradually delivering since February, and AstraZeneca which will deliver in June.
He also criticized private hospitals, which have banded together to demand liberalization of vaccine rules so that they can start treating the many patients who have been asking to be inoculated.
“We already met with them and they understood,” he said. “However, they did not admit they had been wrong in thinking they could buy [vaccines] from anywhere but the government prohibited them from doing so. This criticism persists.”
He also mentioned a case in which a hospital advertised it would accept bookings for a certain vaccine.
“It broke the FDA law because it advertised what it didn’t have in hand,” he said. “After it was fined, it remained silent, further fueling the misunderstanding that the government has been an obstacle.”
Source: CTN News, Asian Times, Bangkok Post, Reuters