Health
Thai FDA Approves Pfizer COVID-19 Vaccine For Children Aged 6 Months -5 Years
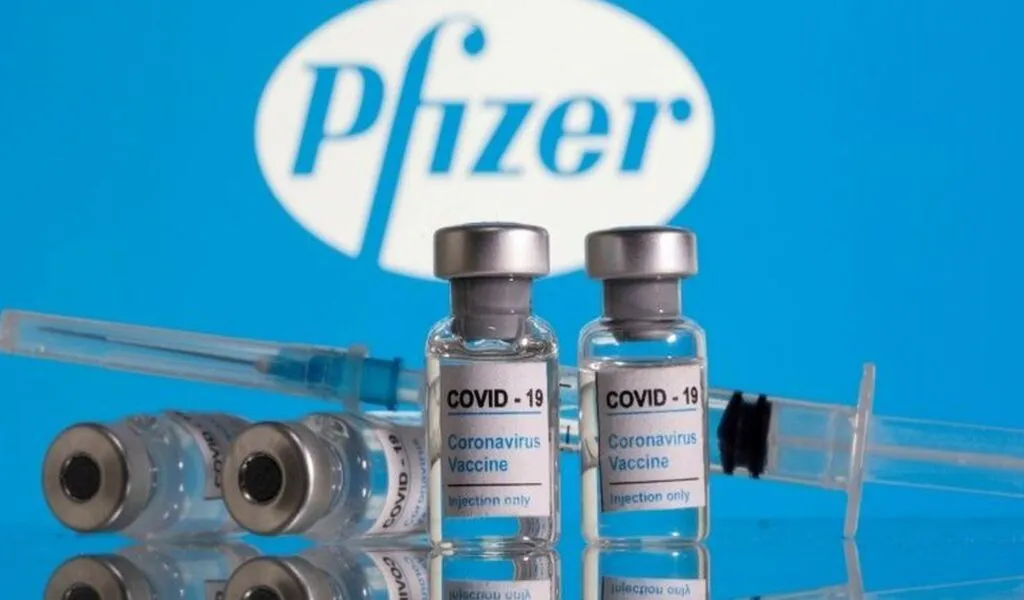
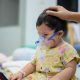
(CTN News) – Pfizer’s COVID-19 vaccine in maroon caps is now approved for use in children aged 6 months to 5 years, with three injections of 3g each.
In terms of COVID-19 prevention, the vaccine is 80.3% effective.
TFDA’s Secretary-General, Dr. Paisarn Dunkum, informed that on August 23.
Yesterday (Wednesday), Dr. Paisarn Dunkum, the Secretary-General of Thailand’s Food and Drug Administration (TFDA), informed me that, on August 23rd.
A subcommittee of the TFDA that was considering registering modern pharmacopeia for humans as a vaccine against COVID-19 approved the expansion of Pfizer’s Comirnaty vaccine to include children aged 6 months to 5 years old.
People aged 12 and older had previously been allowed to use the vaccine for auto-immune stimulation. The age range was extended to children aged 5 to 11 years old.
Children received two doses of intramuscular vaccination 21 days apart, using a 10g dose of the vaccine.
In children aged 6 months to 5 years, 3g of the vaccine will be injected in three injections. After the first injection, the second follows 3 weeks later, and the third 8 weeks later.
Thailand has vaccinated more than 57 million people against COVID-19, of whom 31.6 million have received booster shots.
Related CTN News:
-
Oral Polio Vaccine Linked to Thousands of Polio Cases Since 2017
-
US to Provide Extra 50,000 Monkeypox Vaccines For Pride by Gay Men