Health
Pfizer Says its Pill Paxlovid is 89% Effective Against Covid-19
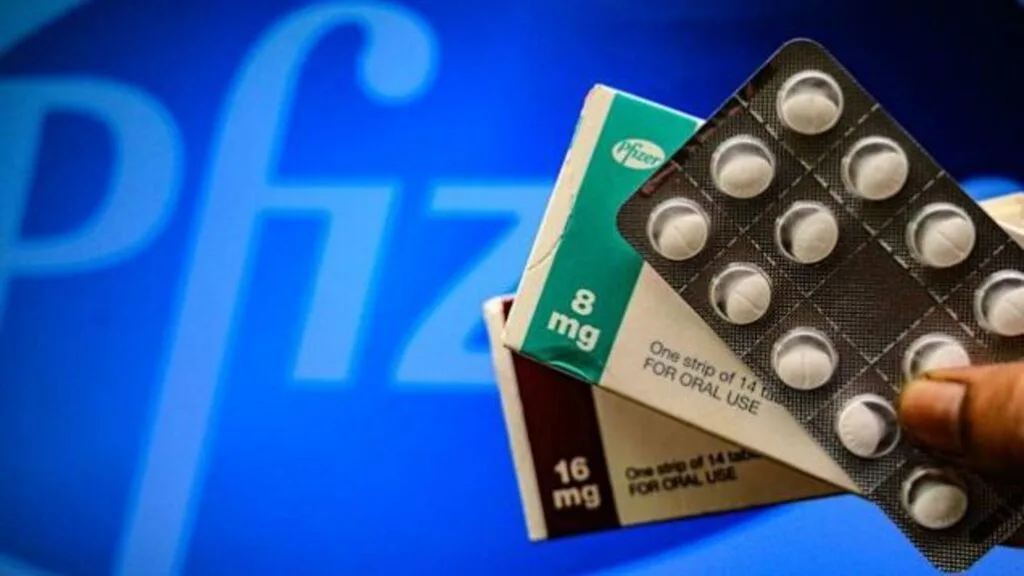
American multinational pharmaceutical corporation Pfizer said Friday that a clinical trial of its Covid-19 pill had shown it is highly effective. The Pfizer drug Paxlovid achieved an 89% reduction in risk of hospitalization or death, Pfizer CEO Albert Bourla said.
Pfizer’s Paxlovid is the second anti-Covid-19 pill after that of Merck, which is actually an influenza medicine rebranded to fight the coronavirus. Pfizer said its medication had been created specifically to fight Covid-19.
The drug achieved an 89% reduction in risk of hospitalization or death among adult patients with Covid-19. Patients who are at high risk of progressing to severe illness, the spokesperson said.
The results from the middle- to late-stage clinical trial were so strong that Pfizer will stop recruiting new people for the trial, Bourla said. The news about Paxlovid is a real game-changer in the global efforts to halt the devastation from the coronavirus.
Pfizer will submit the data to the US Food and Drug Administration as soon as possible for Emergency Use Authorisation. The company started developing Paxlovid in March 2020.
Paxlovid also has the potential to save patients’ lives and reduce the severity of Covid-19 infections. It can also eliminate up to nine out of 10 hospitalizations.
The United Kingdom on Thursday became the first country to approve an anti-Covid-19 pill. The British government green-lighted the use of molnupiravir to treat patients suffering from mild to moderate covid-19 infections.
Paxlovid is known as a “protease inhibitor” and has been shown in lab tests to jam up the virus’ replication abilities. Should it work in real life, it will likely only be effective at the early stages of the covid-19 infection.
Related News:
12-Year-old Girl Becomes Severely ill after Pfizer Vaccine Shot
Thai Government Condemns Genocide Claim Over Pfizer Vaccinations