Health
Pfizer and BioNTech Seek U.S. Authorization For Vaccine Booster Retooled For Omicron
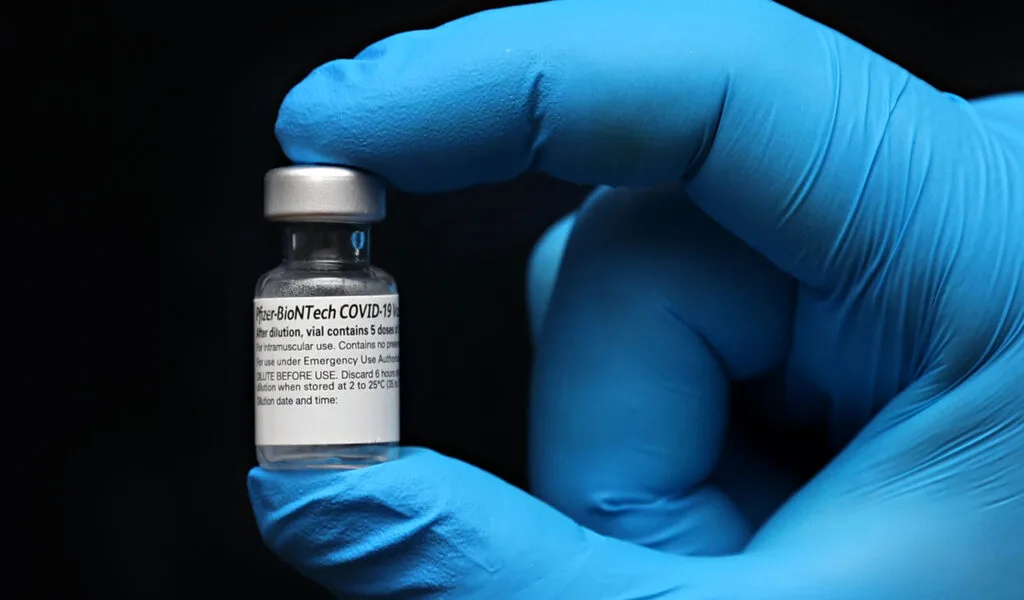
(CTN News) – On Monday, Pfizer Inc. (PFE.N) and German partner BioNTech said they had sought U.S. approval for a COVID-19 vaccine booster retooled to target the Omicron variant and that doses would be available immediately after approval.
The FDA was requested to approve a so-called bivalent vaccine containing the dominant BA.4 and BA.5 variants of the virus along with the original coronavirus strain. Ages 12 and up are recommended.
As part of a $3.2 billion deal with the U.S. government, Pfizer is ready to deliver 105 million doses in September, including Omicron-tailored injections.
We have rapidly scaled up production and are ready to begin distributing bivalent Omicron BA.4/BA.5 boosters if authorized,” Pfizer Chief Executive Albert Bourla said.
Vaccination campaigns are being prepared for the fall in the UK, the United States, and EU members. Last week, Britain cleared a bivalent vaccine by Moderna (MRNA.O).
Bourla said Pfizer will complete its submission to the European Medicines Agency soon.
COVID-19 vaccine makers were asked in June to tailor shots to target the two subvariants, and the FDA said it would not require new human testing for approval, as with flu vaccines.
In a mid-to-late stage study, Pfizer’s older BA.1-tailored vaccine generated a superior immune response against the subvariant than the original shot.
The BA.4/BA.5 vaccine will be studied in people aged 12 and older this month.
More In Health
Related CTN News:
-
10 Early Signs Of Alzheimer’s And Dementia That Need To Be Recognized
-
Monkeypox Vaccine: Can It Stop The Current Outbreak?
-
U.K. Approves New Moderna Vaccine Targeting Omicron Variant