Covid-19
Moderna Says Bivalent Vaccine Appears To Work Against Omicron Subvariants BA.4 And BA.5
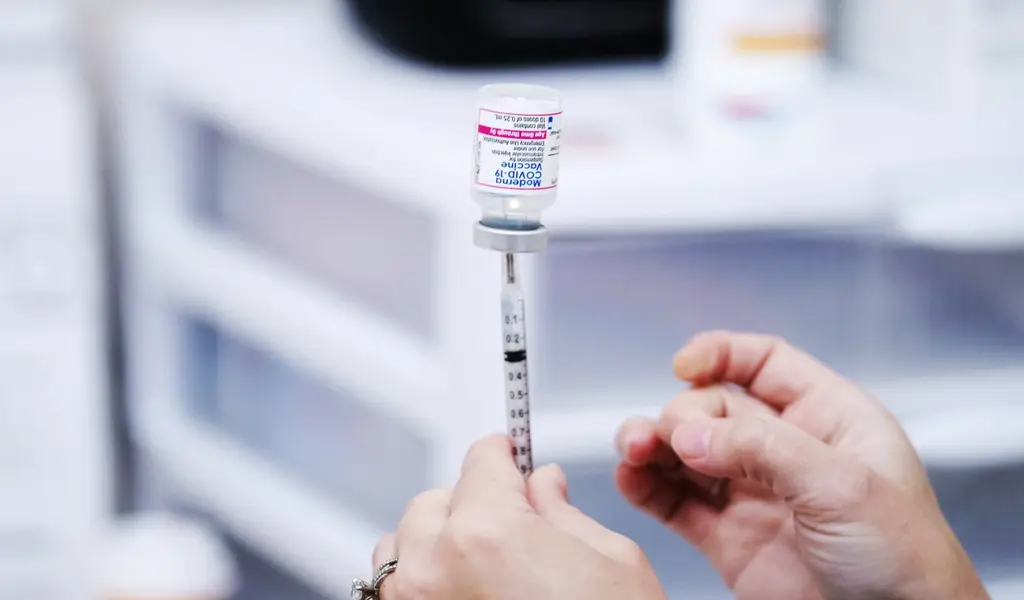
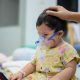
(CTN News) – Moderna said in a news release Wednesday that its updated Covid-19 booster shot appears to work against the fast-spreading omicron subvariants BA.4 and BA.5.
Moderna has said that it hopes the bivalent vaccine will be approved for use in the United States this fall. The vaccine targets both the original omicron variant and the original Coronavirus strain simultaneously.
The updated shot elicited about a fivefold increase in antibodies against BA.4 and BA.5 in previously vaccinated and infected individuals, according to the company.
According to the company, the shot elicited about threefold fewer antibodies against the two subvariants than against the original omicron variant.
Researchers at Moderna said the findings add to findings shared earlier this month, which found a 50 microgram dose of the new shot – the same dosage as the current booster shot – provided strong protection against the omicron strain.
The full data has not yet been made available to outside scientists for analysis, as Moderna announced its findings in a news release.
Experts are concerned about BA.4 and BA.5′s ability to dodge immunity and cause more reinfections in the U.S. In addition, they may also cause more severe illnesses.
According to Dr. William Schaffner, an infectious diseases expert at Vanderbilt University Medical Center, Moderna’s trial results are “good news,” since the updated shot should protect against BA.4 and BA.5, which are expected to dominate the country by summer.
The shot, designed to target the original variant, had been unsure whether it would work against BA.4 and BA.5. About 35% of all new Covid cases are caused by the two subvariants.
Related CTN News: